BioAcyl Corp

BioAcyl Corp |
 |
| Resource type: Journal Article BibTeX citation key: Ogharandukun2020a View all bibliographic details |
Categories: BioAcyl Corp Subcategories: Glycobiology Creators: Damtae, Ogharandukun, others, Tewolde Collection: bioRxiv |
Views: 5/235
|
| Abstract |
|
The surfaces of cells and pathogens are covered with short polymers of sugars known as glycans. Complex N-glycans have a core of three mannose sugars, with distal repeats of N-acetylglucosamine and galactose sugars terminating with sialic acid (SA). Long-range slime-like and short-range Velcro-like self-adhesions were observed between SA and mannose residues, respectively, in ill-defined monolayers. We investigated if and how these adhesions translate when SA and mannose residues are presented in complex N-glycan shields on two pseudo-typed viruses brought together in force spectroscopy (FS). Slime-like adhesions were observed between the shields at higher ramp rates, whereas Velcro-like adhesions were observed at lower rates. The complex glycan shield appears penetrable at the lower ramp rates allowing the adhesion from the mannose core to be accessed; whereas the whole virus appears compressed at higher rates permitting only surface SA adhesions to be sampled. The slime-like and velcro-like adhesions were lost when SA and mannose, respectively, were cleaved with glycosidases. While virus self-adhesion in FS was modulated by glycan penetrability, virus self-aggregation in solution was only determined by the surface sugar. Mannose-terminal viruses self-aggregated in solution, while SA-terminal ones required Ca2+ ions to self-aggregate. Viruses with galactose or N-acetylglucosamine surfaces did not self-aggregate, irrespective of whether or not a mannose core was present below the N-acetylglucosamine surface. Well-defined rules appear to govern the self-adhesion and -aggregation of N-glycosylated surfaces, regardless of whether the sugars are presented in ill-defined monolayer, or N-glycan, or even polymer architecture.
|
| Notes |
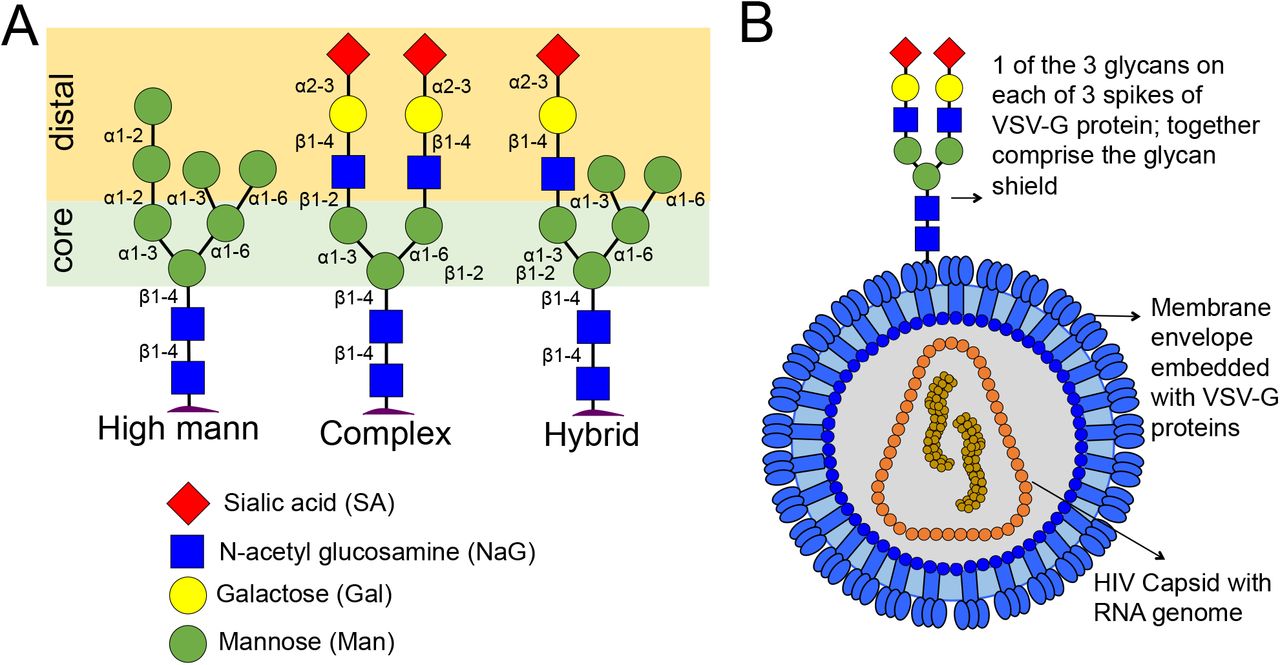
Types of N-glycans protruding from proteins in cell membranes. [B] Presentation of complex N-glycans within the glycan shields of HIV-1 virus pseudotyped with VSV-G proteins. |